On Sept. 16, 1987, policymakers and scientists from around the world gathered at the International Civil Aviation Organization’s headquarters in Montreal, preparing to take action on the day’s most urgent topic: Depletion of the Earth’s protective ozone layer.
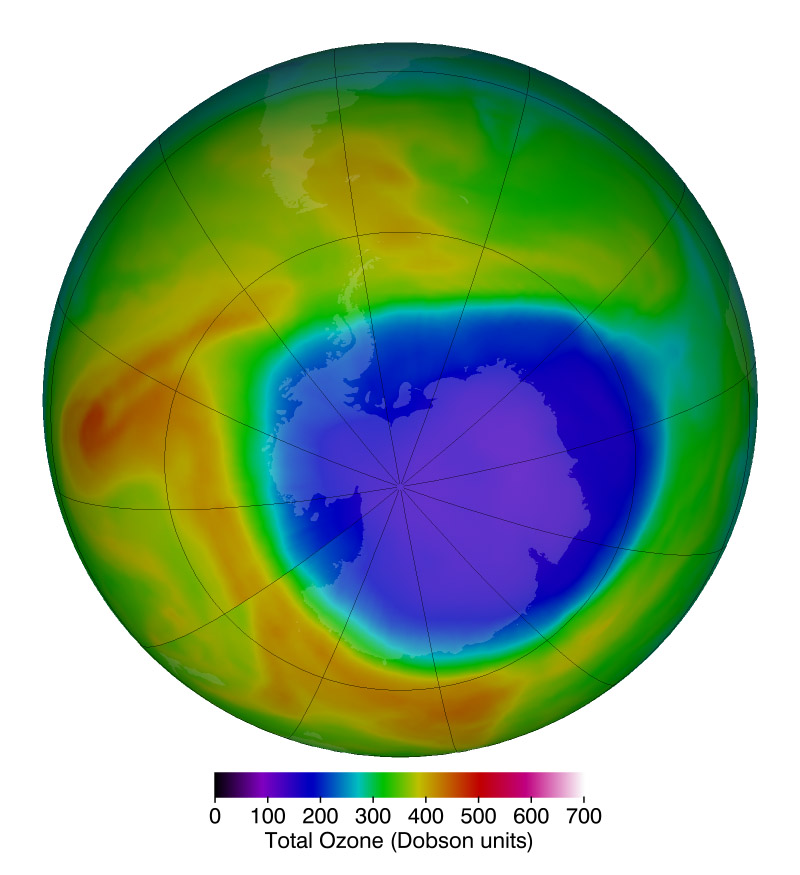
Two years before, researchers from the British Antarctic Survey had stunned the world with the first paper demonstrating that atmospheric ozone levels over Antarctica were dropping at an astonishing rate during the southern hemisphere spring. Shortly after the British paper, NASA showed images from its Total Ozone Mapping Spectrometer (TOMS) that not only confirmed the falling ozone levels, but also showed the extent was broader than anyone realized. The “ozone hole,” as the severely depleted region was dubbed, was the size of the entire Antarctic continent.
Some scientists had warned since the 1970’s that chemicals called chlorofluorocarbons (CFCs) posed a threat to the ozone layer, but no one knew for sure what was causing the ozone hole to develop. The discovery lent urgency to the discussion: How could the world repair the ozone layer before it was too late?
Ozone – a chemical made of three oxygen atoms – is mostly found in a layer about 8-30 miles above Earth’s surface, in the stratosphere. It absorbs harmful ultraviolet (UV) radiation from the Sun, shielding plants, animals and humans from damage ranging from crop death to skin cancer.
“If there were no ozone layer, the Sun would sterilize Earth’s surface,” said Paul Newman, chief scientist for Earth Sciences at NASA’s Goddard Space Flight Center in Greenbelt, Maryland.
On September 16, 1987, Newman was a young atmospheric scientist at Goddard, analyzing data coming back from the Antarctic Airborne Ozone Expedition (AAOE) – where another NASA scientist, Susan Strahan, stood with her colleagues looking at a bulletin board in Punta Arenas, Chile. Strahan analyzed atmospheric chemistry data from the sleek, long-winged ER-2 plane flying into the Antarctic polar vortex to measure ozone and chemicals that could react with it.
That day’s data would yield the famous “smoking gun plot”: The data showing that as a chemical called chlorine monoxide increased in the Antarctic stratosphere, ozone decreased. Chlorine monoxide was known to be present in the atmosphere, but had previously been observed only at lower concentrations than the AAOE team measured — these levels came from a complex set of chemical reactions occurring in the Antarctic following the breakdown of CFCs by UV radiation in the stratosphere. The data disproved other theories and gave scientists evidence that CFCs were causing the ozone hole.
Strahan and her colleagues’ data would not be published until later, but by the end of that day in 1987, twenty-seven nations agreed to the Montreal Protocol on Substances that Deplete the Ozone Layer: “Perhaps the single most successful international agreement to date,” said former United Nations Secretary-General Kofi Annan in 2003. The Montreal Protocol created a timetable for controlling production and consumption of CFCs. Over the next few years, the science of ozone depletion was more firmly established, manufacturers introduced replacement chemicals that were safer for the environment, and the Montreal Protocol was strengthened several times to stop wide-scale production and use of CFCs and related molecules.
The long journey toward recovery had begun.
Today, Newman and Strahan are leaders in atmospheric science and both sit at NASA Goddard: Newman as chief scientist for Earth Sciences and co-chair of the Scientific Assessment Panel (SAP) to the Montreal Protocol, Strahan as a principal scientist for the Universities Space Research Association. And today, they both keep an eye on Earth’s atmosphere, continuing NASA’s long-running research and monitoring efforts on stratospheric ozone (which go back to the 1970s) into the future.
CFCs: Danger at High Altitudes
CFCs were not always the villain in this story. Invented for use as refrigerants in the 1920’s, CFCs represented a technological breakthrough: They were versatile, but more importantly, they were neither toxic nor flammable. Older refrigeration chemicals were lethal if leaked; CFCs did not harm human health or react with other chemicals in the lower atmosphere.
The problem is that while CFCs are inert at the surface, the story changes in the stratosphere.
“CFCs are emitted at the surface. We make a fridge, and the compound leaks out,” said Strahan. “The emissions start in the troposphere (the atmospheric layer closest to Earth’s surface) and work their way up to the stratosphere.”
Once CFCs diffuse above the protection of the ozone layer, UV radiation breaks them apart, releasing highly reactive chlorine atoms. At first, these react with other chemicals to create hydrochloric acid and chlorine nitrate — called “reservoir gases,” Strahan said, because they typically store chlorine in stable molecules.
But the polar regions support chemical reactions that could not happen anywhere else on Earth. The intense cold of polar winters allows the formation of thin clouds, despite low atmospheric moisture. And the polar vortex winds encircle the Antarctic region, trapping the chemicals within its boundary. Hydrochloric acid and chlorine nitrate react on the surfaces of these the thin cloud particles to free the reactive chlorine once again, and when the Sun returns in the spring, the UV radiation initiates the catalytic chlorine-ozone reactions that destroy the ozone layer. One chlorine atom can destroy thousands of ozone molecules – and with millions of tons of CFCs pumped into the atmosphere from the 1920’s through the early 1990’s, the Antarctic polar region bore the brunt of the damage.
“If we had done nothing, if the Montreal Protocol had not been signed, by this time things would have been quite disastrous,” said Newman. “Ozone levels would be way down; UV levels would be way up. Because of increased UV radiation at the surface, we would have had global crop losses, people would sunburn faster and skin cancer would be going up. Food prices would shoot up; the poor people of the world would have greatly suffered.”

First Steps to Recovery
Today, 33 years later, the ozone hole is showing its first signs of recovery. Strahan and her colleague Anne Douglass published one of the first studies in 2018 confirming that atmospheric chlorine levels are falling in step with reduced ozone depletion over Antarctica — proof that the Montreal Protocol is working.
These first hopeful signs represent a global success story: Policymakers, scientists and companies around the world joined forces to find a solution to an urgent problem. Much of the data that empowered these decisions came from NASA scientists and instruments. Ongoing ground- and space-based monitoring of ozone and other trace gases, by NASA and other institutions, will help inform development of environmental policies designed to make sure levels continue trending in a positive direction even in the midst of other changes, such as Earth’s warming climate.
“If you don’t know how much ozone is up there, you don’t know if it’s getting better or worse,” said Strahan. “If it does change, was it natural variability or was it caused by humans? Having a long data record of ozone and other gases directly related to its chemistry is really important.”
Credits: NASA’s Goddard Space Flight Center/Katy Mersmann
Download this and supporting imagery from NASA Goddard’s Scientific Visualization Studio
Today, NASA monitors ozone from space using the Microwave Limb Sounder (MLS) and Ozone Monitoring Instrument aboard its Aura spacecraft, and the MLS also measures trace gases containing chlorine. Strahan and Douglass’ 2018 study used MLS measurements of hydrochloric acid, a form that chlorine takes after destroying ozone, to calculate total stratospheric inorganic chlorine above Antarctica. Inorganic chlorine compounds like hydrochloric acid have no carbon molecules, which allows researchers to differentiate between them and chlorine still tied up in CFCs.
Additionally, the Stratospheric Aerosol and Gas Experiment III measures ozone and trace gases from its vantage point aboard the International Space Station, and the NASA-NOAA Ozone Mapping Profiler Suite aboard the Suomi-NPP satellite measures both total column ozone and ozone profiles. These instruments had precursors on earlier NASA satellites, and they — along with space, air and ground measurements from partner organizations like the National Oceanic and Atmospheric Administration and global partners — will help scientists keep track of the ozone hole’s recovery.
“When it comes to a clear sign the ozone hole is going away, it could still be a couple decades before we can look up and say it’s smaller every single year than it was in the early 2000’s,” Strahan said. “Most years since then, it’s been a bit smaller, but occasionally we’ll have a really cold year and a big hole again. We’re going to have that kind of variability going forward, but once we get to 2040 or so, there will be so much less chlorine that the holes will be smaller even in cold years. It will be a long, bumpy road, but we’re headed in the right direction. We just need to be patient and keep up the good work.”
Banner Image: Ozone – a chemical made of three oxygen atoms – is mostly found in a layer about 8-30 miles above Earth’s surface, in the stratosphere. It absorbs harmful ultraviolet (UV) radiation from the Sun, shielding plants, animals and humans from damage ranging from crop death to skin cancer. Credit: NASA / Stuart A. Snodgrass
By Jessica Merzdorf
NASA’s Goddard Space Flight Center, Greenbelt, Maryland